Methionine is a sulfur-containing amino acid that is essential in humans. While it’s one of the rarest amino acids in proteins, its importance cannot be overstated as it is used in a range of biological processes including metal coordination, antioxidant stress, and aging. Despite its importance, it has not seen nearly as much attention as the other sulfur-containing amino acid, cysteine. Luckily, in recent years, new methionine selective bioconjugation methods have been developed, allowing for insight into its activity in the body.
Some examples of methionine selective bioconjugation methods include conjugation to antibodies via ReACT chemistry, conjugation to polymers using Poly-MPC chemistry, and passive bioconjugation to citrate capped surfaces.
Reasons for Methionine Selective Bioconjugation of Proteins
Since methionine is so rare in proteins, bioconjugation is an excellent strategy to understand its activity in the body. The uniqueness of methionine also makes it an excellent target for studies on methionine-containing proteins. Bioconjugation is a technique to couple a biomolecule to another molecule. By coupling methionine-containing proteins to other molecules, you can gain access to a range of uses.
There are many reasons why a scientist may want to bioconjugate to methionine containing proteins including: antibody studies, creation of novel biomaterials using polymers, surface binding in biosensors, and protein-DNA bioconjugates for genetic studies.
Bioconjugation of Methionine-Containing Proteins to Antibodies
Methionine-containing proteins bioconjugated to antibodies have been used in immune studies. Many antibodies contain multiple surface lysines for conjugation meaning that there is often low specificity, while methionine offers a more specific target. Recent studies have shown that specific methionine-containing protein bioconjugation with antibodies can be used for fluorescence studies, affinity labels, and more.
For example, in this study, the authors managed to bioconjugate the methionine residue of a modified antibody to green fluorescent protein (GFP) using the ReACT method (which is discussed later in this article). GFP is used in fluorescence microscopy. By tagging the antibody with GFP, the authors would allow it to be detected and monitored using fluorescence microscopy. The authors noted that this strategy was potentially valuable because it did not result in any background labeling of wild-type antibodies.
Bioconjugation of Methionine-Containing Proteins to Polymers
The use of polymer bioconjugates has seen increasing use in the last two decades. The bioconjugation of proteins to polymers can allow for them to be fixed in place for further modifications or to act as reaction sites for further reactions. This bioconjugation can be passive or active, using passive forces or covalent reactions, respectively.
The authors of this study used poly-MPC chemistry to bioconjugate a phospholipid polymer to a protein while maintaining the conformation of the protein. MPC polymers have seen use in artificial hip joints as a boundary lubricant. In this case, the authors wanted to further study its usefulness as a stabilization material for proteins via bioconjugation. Methionine residues on the protein are reacted with MPC to activate them and then bound to the phospholipid. The conformation of the protein was not affected by binding it to the polymer. The authors found that the conformation was preserved even under heat-quenching stress.
Bioconjugation of Methionine-Containing Proteins to Surfaces
The bioconjugation of proteins to surfaces allows for the development of new assays and potential biosensors. As methionine is specific to certain proteins, methionine-containing proteins can be selectively bound to nanoparticles. These nanoparticles can have different properties such as magnetism or electrostatic properties that can be manipulated to interact with the protein in-situ.
For example, this study used citrate-capped gold nanoparticles with amino acids for sensing applications. The authors measured the passive absorption of the methionine to the gold nanoparticles using UV-Vis spectrophotometry and varying the pH in solution. The passive adsorption of a protein to a nanoparticle varies depending on the experimental conditions.
We’ve also discussed other methods for bioconjugation to surfaces in a related article.
Methionine Assay – How to Detect Methionine Amino Acids in a Protein
Many methionine residues on a protein are hidden within the structure and are therefore inaccessible for bioconjugation reactions. This makes it difficult to target specific proteins without knowing whether they have accessible methionine residues. Sadly, no rapid method for detecting accessible methionine residues in proteins exists.
Methionine assay methods include computation modeling and quantification of methionine sulfoxide using isotopic labeling of unoxidized methionine residues.
Some studies have used computational modeling to determine the number of accessible methionine sites. The authors used software called MODELLER and protein data bank data for an antibody as the template. The software allowed the authors to generate models of the antibody and look at the spatial restraints on each methionine. This is very efficient in terms of resources but requires you to have access to an already modeled protein to perform the computational studies.
Several other studies (for example this, and this) have instead been able to quantify the oxidized methionine residue, methionine sulfoxide. Methionine is subject to reversible oxidation and reduction, so in theory, if you can quantify the methionine sulfoxide instead, you will have an idea of the accessible sites. Their method involved the isotopic labeling of unoxidized methionine residues with 18O hydrogen peroxide, then using LC-MS/MS to measure the ratio of 18O to 16O.
Different Methods for Methionine Selective Bioconjugation
There is a range of different methods available for the selective bioconjugation of methionine to different targets.
Methods for methionine selective bioconjugation include selective bioconjugation of methionine to antibodies using ReACT reactions of the sulfur groups, MPC chemistry for bioconjugation to a polymer, and bioconjugation to surfaces using self-assembly reactions.

Related articles:
- Learn more about orthogonal bioconjugation techniques, tips and tricks in our article
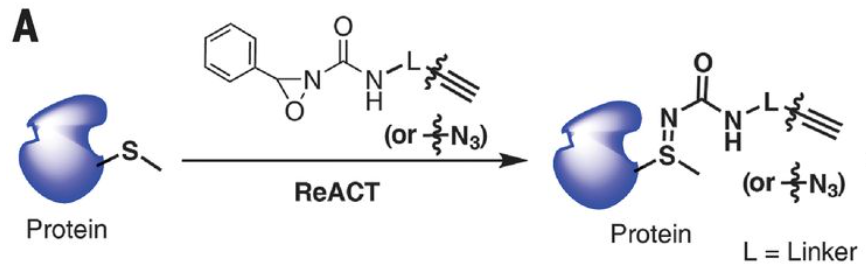
Method for Selective Bioconjugation of a Methionine-Containing Protein to an Antibody
A novel method called the ReACT (redox activated chemical tagging) strategy allows for the selective bioconjugation of methionine proteins to an antibody.
Step 1. Collect the necessary materials.
You’ll need your methionine-containing protein, an antibody (or antibody fragment) functionalized with oxaziridine groups, and methanol.
Step 2. Add your protein and antibody together in methanol.
Carefully add the protein and antibody together in methanol. Stir gently.
Step 3. Monitor the reaction progress using NMR.
You can monitor the progress of the reaction by detecting the chemical shift of the methionine methyl group.
Step 4. Once the reaction has gone to completion, you will have your bioconjugated protein and antibody.
They will be fairly resistant to acidic and basic conditions, but you’ll need to keep them cool to ensure their stability.
Method 2. Methionine Bioconjugation of a Protein to a Polymer
Polymer materials can be designed with functionalized surfaces that allow them to be conjugated to a target such as a protein. For this method, 2-methacryloyloxyethyl phosphorylcholine (MPC) functionalized polymers are used to bioconjugate to bovine serum albumin (BSA).
Step 1. Prepare an MPC functionalized polymer.
Several methods are available online, such as this one.
Step 2. Add your MPC-polymer to a solution containing your target protein.
Your BSA should be stabilized in a phosphate buffer solution. Add the MPC polymer directly to the solution
Step 3. Incubate the reaction for 1 hour.
The incubation should be at room temperature.
Step 4. Lypophilize your conjugate
This will ensure the stability of the protein-polymer conjugate for further studies.
Method 3. Methionine Bioconjugation of a Protein to a Surface
Sigma-Aldrich offers a method for the bioconjugation of the surface of iron nanoparticles to a methionine-containing protein.
Step 1. Gather your reagents, iron nanoparticles, and protein.
You’ll need amine-functionalized nanoparticles, a protein presenting free thiol groups from methionine residues, Sulfo-SMCC, mercaptosuccinic acid, and a PBS Buffer.
Step 2. Add your nanoparticles and sulfo-SMCC into a sample tube containing the PBS buffer.
Your reaction mixture needs to be buffered to ensure that the reaction has the highest possible yield.
Step 3. Stir the mixture for 1 hour.
The iron nanoparticles need time to react with the sulfo-SMCC.
Step 4. Add your target protein to the activated iron nanoparticles. Allow the mixture to stir for 4 hours.
Allow the reaction to proceed at room temperature for 4 hours.
Step 5. Add mercaptosuccinic acid to quench the reaction.
After 4 hours the reaction will need to be quenched to ensure the stability of the bioconjugate.
Step 6. Purify your nanoparticles using a centrifuge.
Centrifuge the particles and remove the supernatant. Resuspend them in PBS to act as a storage buffer. They can be stored in a refrigerator to improve their longevity.
Commercially Available Methionine Bioconjugation Reagents
| Supplier | How does the reagent work | Price in USD |
| Sigma-Aldrich Sulfo-MCC | See method 3 above. | $294 for 50 mg of sulfo-SMCC |
| Thermo-Fisher L-photo-methionine | Incorporated into proteins during synthesis then activated for conjugation reactions. | $426 for 100 mg of L-Photo-Methionine |
| Click Chemistry Tools AHA | Feed to cultured cells and incorporated into proteins. Allows for in-cell labeling. | $115 for 25 mg of AHA |