Thymidine and BrdU, Cell Proliferation Assay Summary
How do you know if your cultured cells are growing? Does your new cancer drug affect cell proliferation? What’s the effect of VEGF on endothelial cells? As you can tell, knowing how to perform cell proliferation assays is an absolutely essential skill for anyone in biology, biochemistry, or pharmaceutics. Radioactive Thymidine cell proliferation assays have been used since for over 40 years to detect whether cells are growing. The principle is simple: cells will incorporate Thymidine into their DNA as they proliferate. However, dealing with radioactivity is painful and annoying, so new fluorescence-based, non-radioactive, BrdU and EdU cell proliferation assays have become the new mainstay technique. These molecules are both thymidine analogs and hence work using the same principle as radioactive thymidine. In today’s guide, we will learn Step-by-Step, the theory behind these assays and how to apply them in the lab. Combining our techniques of MTT Cell Viability assays and Flow Cytometry or FACS, we are really building up a great list of skills to analyze biological phenomena!
Note: If you’re writing research papers, I highly recommend Grammarly – it’s a free grammar check plugin for Chrome. Try it out here…
Principle of Cell proliferation assays with nucleotide analogs
3H-Thymidine is a radioactive version of the Thymine DNA base (thymine + the sugar backbone = thymidine). When cells are incubated with thymidine, they use the radiolabeled thymidine to synthesize DNA and incorporate it into their DNA backbone. So, thymidine is an excellent measure of DNA synthesis in cells that have undergone the S-Phase of cell replication. Similarly, BrdU is a Thymidine analog that lacks the radioactivity from tritium and it is used identically to Thymidine. Just incubate cells in the presence of BrdU. However, unlike radioactive thymidine, BrdU is detected with Anti-BrdU antibodies.
A quick summary picture is shown below.
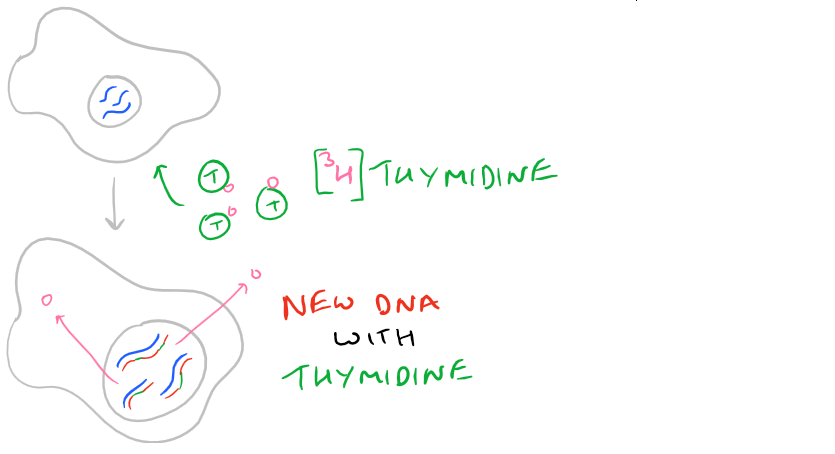
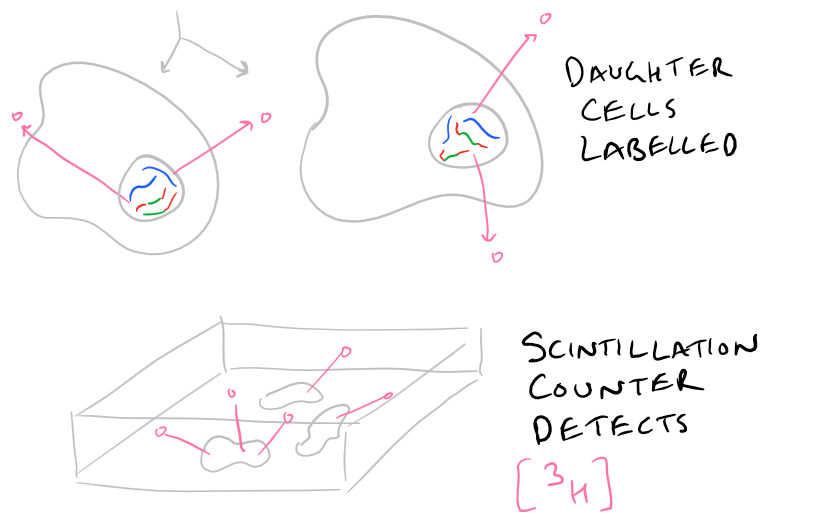
Using Thymidine vs. BrdU. Cell Proliferation Assay Tips and Tricks
Taking things with a grain of salt: Note that DNA replication can happen even when cells are not proliferating. For example, if you have damaged DNA (ie. DNA repair is taking place). So, Thymidine and BrdU assays are really DNA replication assays and not perfect cell proliferation assays. But, for the most part, they are the gold-standard when looking for cell proliferation.
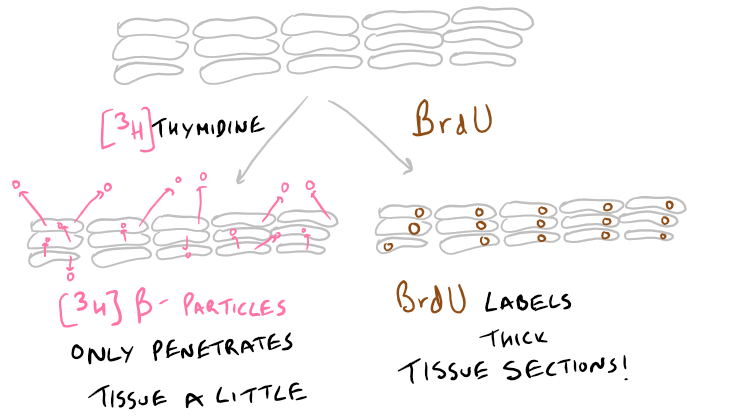
Thick tissue sections? Choose your cell proliferation assay wisely: The 3H-Thymidine assay uses radioactivity. And the beta particles that are generated by this method cannot penetrate very deep into tissue. So, if you’re labeling tissue sections, make sure they are extremely thin! In these cases, BrdU is a great option because it penetrates deep into tissue and can be detected even from 50 um thick slices. This is illustrated below in the picture.
Not enough signal? Add more: Since cells are substituting the radioactive thymidine into their DNA. Adding more thymidine means you’ll get more incorporation. And, more incorporation means you’ll get more signal! So, if your signal is low, just add more of the nucleotide. BrdU, however, doesn’t behave this way. There is a limit at which adding BrdU doesn’t increase your signal.
Want to preserve your tissue? Use 3H-Thymidine: BrdU immunohistochemistry requires you to digest and disrupt tissue for visualization because the antibodies that detect BrdU need to access all the tissue. Detecting radioactivity just needs you to use a scintillation counter.
Other Methods: EdU Cell Proliferation Assay
BrdU immunohistochemistry has the disadvantage that you need antibodies to detect it. Because of this, you need to disrupt the tissue you are staining. EdU is a new version of BrdU which has an azide functional group. This can then easily be detected with a “click” fluorophore. (We’ve discussed more ‘click’ methods in our article on orthogonal bioconjugation techniques). This is shown below:
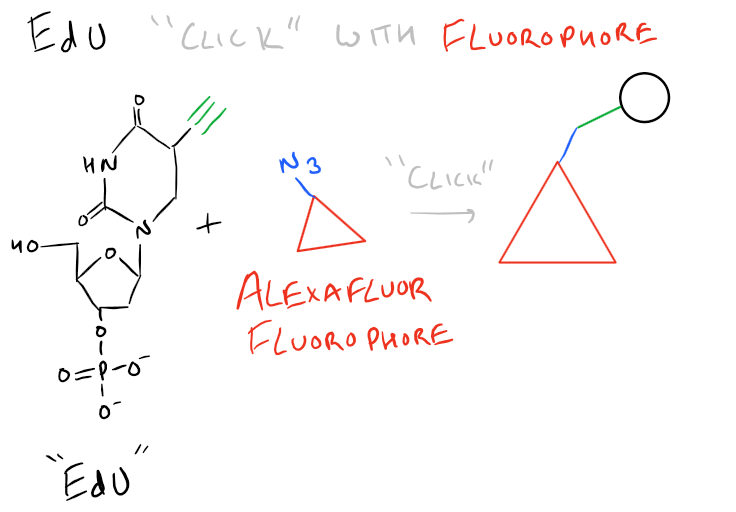
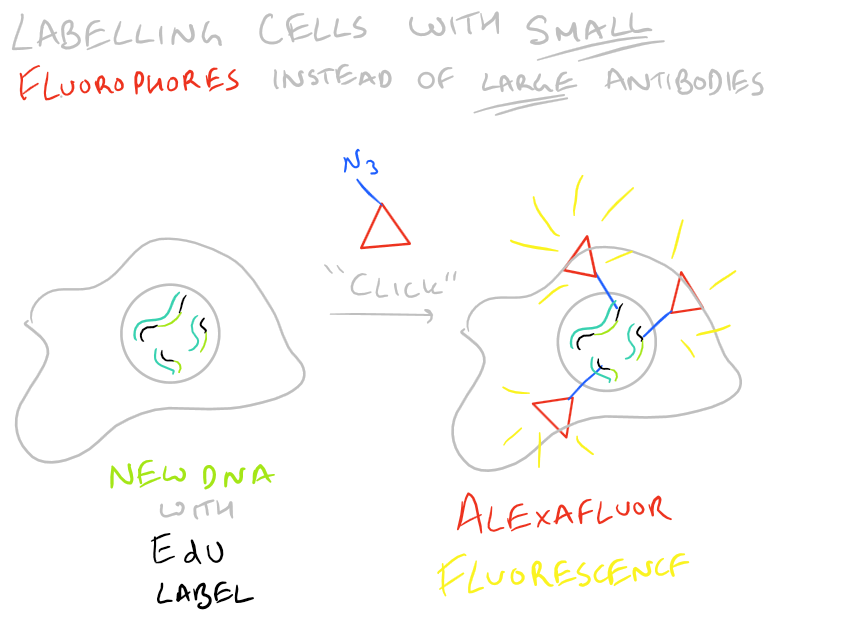
Step by Step Guide to BrdU Cell Proliferation assay in vivo
Here is how you can label proliferating cells in a mouse
Materials for BrdU Assay
BrdU labeling reagent (Invitrogen, #000103)
BrdU staining kit (Invitrogen, #933943)
Opaque dark container for storing stained tissue
PBS
16% Paraformaldehyde in Water (#47608, Sigma Aldrich)
4% Paraformaldehyde
1% Paraformaldehyde with 0.5 M EDTA, pH 8 [Demineralization solution]
Histo-clear (#50-329-51 Fisher Scientific)
Ethanol 100%
Ethanol 95%
Ethanol 70%
Water
Slides
Coverslips
30% Hydrogen Peroxide (H2O2) in Water
Methanol
Petri dish with wet paper towels and paperclips for humidifying tissue (see image)
Note: Grammarly is a free grammar check plugin for Chrome. I used it for this article and really like it! Try it out here…
Step-by-Step Protocol for Cell Proliferation Assay
You’ll want to refer to our Immunofluorescence microscopy guide and our Immunoprecipitation / CoIP Guide to understand this section better. Especially the bit about biotinylated antibodies and their detection.
- Give mice 100 ul of the BrdU labeling agent per 10 g of mouse weight. I.P
- Wait 2-4 hours for labeling the skeletal tissue
- Asphyxiate mice using CO2
- Dissect mouse body parts and rinse with 1x PBS
- Fix mouse tissue in 4% paraformaldehyde solution for 48 h at 4oC
- Demineralize tissue with demineralization solution for 3 weeks. Change solution 1x per day, everyday.
- Rinse sample tissues 3x with PBS for 60 minutes each wash.
- Embed the tissues in paraffin and mount on slides. Make sure sections are less than 20 um thick.
- Air dry the frozen sections on the bench for 1 h.
- Remove paraffin by dipping the slides in Histo-Clear and leaving them for 5 minutes. Repeat this once again.
- Rehydrate sections by dipping in 100% Ethanol for 2 min, then 95% for 2 minutes, then 70% for 2 minutes
- Wash sections 3x with 1x PBS for 2 min at a time.
- Quench endogenous peroxidase activity by submerging sections in 10% H2O2 in methanol for 10 min
- Rinse 3x with PBS for 2 min each.
- Make trypsin solution according to BrdU Staining kit and cover tissue sections.
- Rinse with PBS 3x for 2 min each.
- Add DNA denaturing solution and cover tissue for 30 min.
- Rinse off excess with PBS wash 3x for 2 min each. Then remove the PBS by blotting with tissue paper around the edges of the tissue.
- Add the blocking solution and submerge tissue.
- Add the biotinylated mouse anti-BrdU antibody and incubate for 60 min in the humid chamber petri dish.
- Rinse 3x with PBS
- Add the streptavidin-peroxidase solution. Incubate for 30 min.
- Rinse 3x with PBS
- Freshly make the peroxidase staining solution from the BrdU staining kit. Incubate sections for 5 min in this.
- Add hematoxylin staining solution for 1 min. Not more!.
- Quickly rinse with PBS 2 times for 30 secs.
- Dehydrate slides by incubating in 70% ethanol for 1 min. Then incubate in 95% ethanol for 1 min. Then incubate in 100% ethanol.
- Add Histomount media and put a coverslip on top.
- Use a normal brightfield microscope to image the cells. Make sure to check multiple vieweing areas to get a representative sample of your tissue.
Notes on this BrdU Cell Proliferation Assay Methodology
- Demineralization finishes when the tissue is pliable and can be bent without breaking.
- Embedding tissues in Paraffin will be discussed later in another post.
- After adding peroxidase staining solution, the tissue will become visibly browner
- Staining with hematoxylin will make sections too blue and harder to analyze. Don’t leave in there for more than 1 minute!
- Normal cells that are dividing will have brown nuclei. Non dividing cells will have blue nuclei. % Proliferation = dividing cells / (dividing cells+non dividing cells).
Checking Cell Proliferation in Zebrafish: Video
Applications of BrdU, EdU, and Thymidine Assays on SciGine
BrdU Immunofluorescence Cell Proliferation Assay
In Vivo BrdU Assay for Cell Proliferation of Chondrocytes
Thymocyte proliferation measured using [H]Thymidine
EdU Cell Proliferation Assay with Flow Cytometry
Lamprey Cell Proliferation using EdU Assay
References
Calculation of Cell Proliferation using Thymidine
Duque et al.: How Proliferation assays affect cell behavior
Mead et al.: How to use BrdU with Skeletal tissue sections